 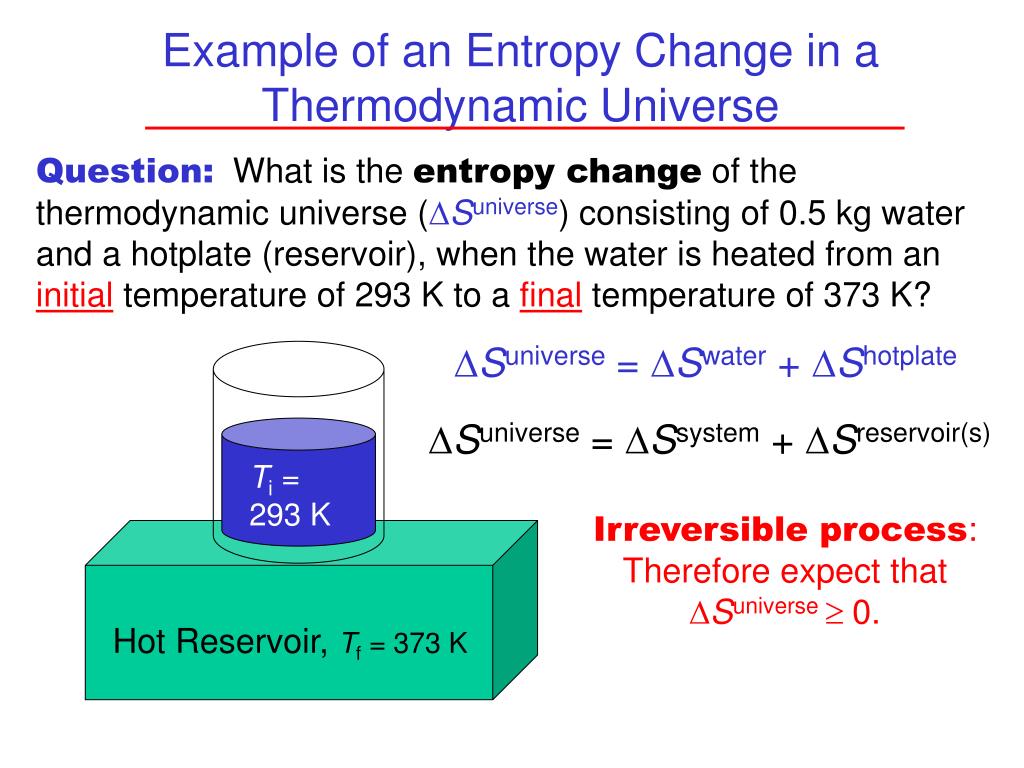 
Here we further explore the nature of this state function and define it mathematically. Calculate the entropy change for 1. 
The enthalpy of fusion for water is 6.01 kJ/mol. In Chapter 13, we introduced the concept of entropy in relation to solution formation. The entropy change for a phase change at constant pressure is given by. To help explain why these phenomena proceed spontaneously in only one direction requires an additional state function called entropy (S), a thermodynamic property of all substances that is proportional to their degree of "disorder". Moreover, the molecules of a gas remain evenly distributed throughout the entire volume of a glass bulb and never spontaneously assemble in only one portion of the available volume. On the basis of determining the entropy change associated with phase transitions is the third law of thermodynamics: the entropy of a perfect crystalline. For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. (2) Optimize the geometry of this conformer with DFT, compute the Hessian matrix from the DFT structure and use the HO vibrational frequencies to calculate S msRRHO. Large-scale calculations of gas phase thermochemistry: Enthalpy of formation, standard. The few simple steps required for the calculation of the absolute entropy are (1) Run CREST in default mode on a starting structure to find the lowest conformer. Entropy is a scientific concept that is most commonly associated with a state of disorder, randomness, or uncertainty. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. Approaches that compute the absolute entropy can be roughly. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush. By the formula of entropy change.\): An Endothermic Reaction. For perfectly crystalline substance entropy S = 0 at 0 KĪbsolute entropy of a pure substance : Absolute value of entropy (unlike absolute value of enthalpy) can be calculated at any temperature. To obtain some idea of what entropy is, it is helpful to imagine what happens when a small quantity of energy is supplied to a very small system. Pure corresponds to no mixing of foriegn substances. For example, the standard entropy of graphite is 6 J K-1 mol-1, whereas that for water it is 70 J K-1 mol-1, and for nitrogen it is 192 J K-1 mol-1. Kinetic energy is directly proportional to Temperature, when temperature becomes zero then kinetic energy will also be zero. There are a wide variety of crystallographic defects. A perfect crystal is a crystal that contains no point, line, or planar defects. Third law of thermodynamics : Entropy of pure and perfectly crystalline substance at 0 K is zero. JEE Main 2022 Question Paper Live DiscussionĪccording to third law of thermodynamics absolute value of entropy can be caculated at any temperatures.Q is positive for energy transferred into the system by heat and negative for energy transferred out of the system by heat. S Q T, where Q is the heat that transfers energy during a process, and T is the absolute temperature at which the process takes place. Difference Between Selling And Marketing The equation for the change in entropy, S, is.TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10 The absolute entropy of a substance at any temperature above 0 K must be determined by calculating the increments of heat q required to bring the substance from 0 K to the temperature of interest, and then summing the ratios q/T.NCERT Solutions For Class 6 Social Science.In this section, we examine two different ways to calculate S for a reaction or a physical change. In contrast, other thermodynamic properties, such as internal energy and enthalpy, can be evaluated in only relative terms, not absolute terms. NCERT Solutions for Class 7 Social Science The area under the curve between 0 K and any temperature T is the absolute entropy of the substance at T. Absolute entropy increases steadily with increasing temperature until the melting point is reached, where it jumps suddenly as the substance undergoes a phase. 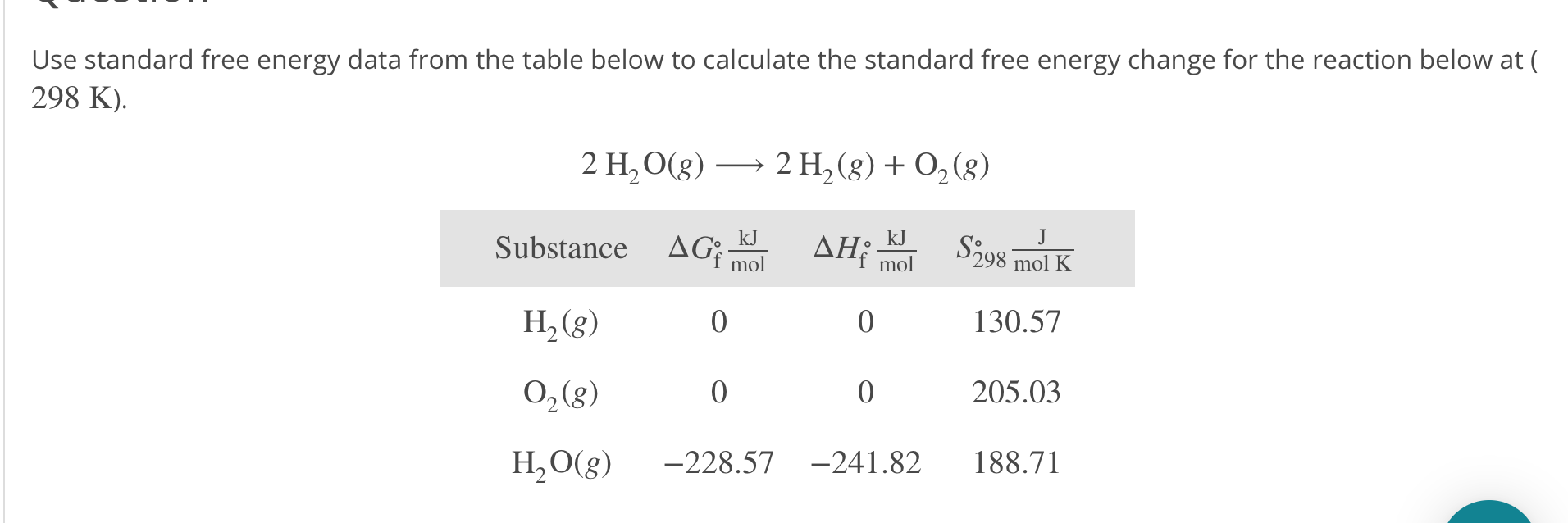
NCERT Solutions for Class 8 Social Science. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
